Handling Blood Samples – 5 Good Practices
While peripheral blood and its precious sub-components are gaining growing attention from the biomedical research field, an increasing body of evidence 1,2 shows that even small changes in the pre-analytical conditions may affect sample quality and integrity. These observations drive the urgent need for establishing good practices and unified protocols for blood sample collection, transportation, processing, and conservation across research centers and hospitals.
Here, we will highlight some of the key good practices to follow when planning your blood collection study according to the analyte of interest.
1. Choose the appropriate blood collection tube for your desired specimen
The main goal of each biospecimen is to fit its analytical purpose. Therefore, in the context of whole blood samples, the blood component of interest will define the collection parameters starting with the choice of blood collection tube. Blood containers carry different lyophilized or soluble additives. The two main types of additives are either clot formation factors for serum retrieval or anti-coagulation agents used for plasma and cell isolation.
Anticoagulants include EDTA, heparin, and sodium citrate. Each one of them is impacting the blood sample, especially in terms of protein profile. Therefore, you should choose the type of anticoagulant according to your downstream applications. For example, avoid heparin-containing tubes for sequencing and genomic studies. But if you are interested in isolating viable PBMCs, then heparin and citrate tubes are the container of choice instead of EDTA-coated tubes3.
Storing containers according to the manufacturer's instructions, at room temperature, and protected from direct sunlight will ensure the good preservation of the additives. Always use blood containers with a valid expiration date.
Keep in mind that you should also consult the type of blood collection tube when using biobanked retrospective collections as samples collected in different containers are likely to generate biased results.
2. Hadling blood samples correctly is crucial during pre-analytical research phase
Standardization of post blood withdrawal handling steps is crucial for ensuring sample quality and integrity preservation. Many efforts are being implemented to secure the uniformity and usability of both retrospective and prospective blood samples for relevant downstream molecular analysis.
An example of such action is the Standard Operating Procedure Integration Working Group at the National Cancer Institute (NCI) which has established Standard Operating Procedures (SOPs) for blood collections designated for plasma and serum isolation.
The pre-analytical stage includes the transportation and storage time between collection and potential pre-analytical processing. The main parameters to be considered are time and temperature. The established temperature for transportation or storage during this period varies between 4-5°C to 18-22°C, depending on the analyte to be isolated 3,4. Initiate downstream blood sample processing within 24hr after collection.
Another important and often disregarded point is the up and down inverting of the blood in the blood container, allowing for the mixing of the blood and additive. This is crucial in the case of plasma and cell isolation as it will ensure the prevention of clot formation, especially in situations involving long transportation steps. Mixing must always be done slowly in an up and down controlled motion. Avoid vigorous shaking as it could damage or activate blood cells and cause hemolysis.
Although these parameters might change between protocols and studies, the important thing is to ensure the manipulation of all samples from one collection follows the established SOP or protocol.
3. Temperature matters all along the way
Temperature is a crucial factor from collection to transportation, isolation, and storage, and it depends on the blood component of interest.
Clot formation naturally occurs at ambient temperature. Cells also require ambient temperature for optimal isolation and viability, and it is vital to make sure this is maintained during winter or summer when temperature significantly fluctuates.
Protein stability and activity are temperature-sensitive and dependent. Therefore, if plasma proteins are the molecule of interest, apply ice packs during transportation and processing steps. It is crucial to ensure that the blood tubes are not in direct contact with the ice because it might lead to hemolysis and cell damage.
Balancing the temperature parameter during simultaneous plasma and cell isolation from the same tube is a challenging task. In such a scenario, it is best to limit transportation time as much as possible and perform faster processing to minimize damage to all blood components.
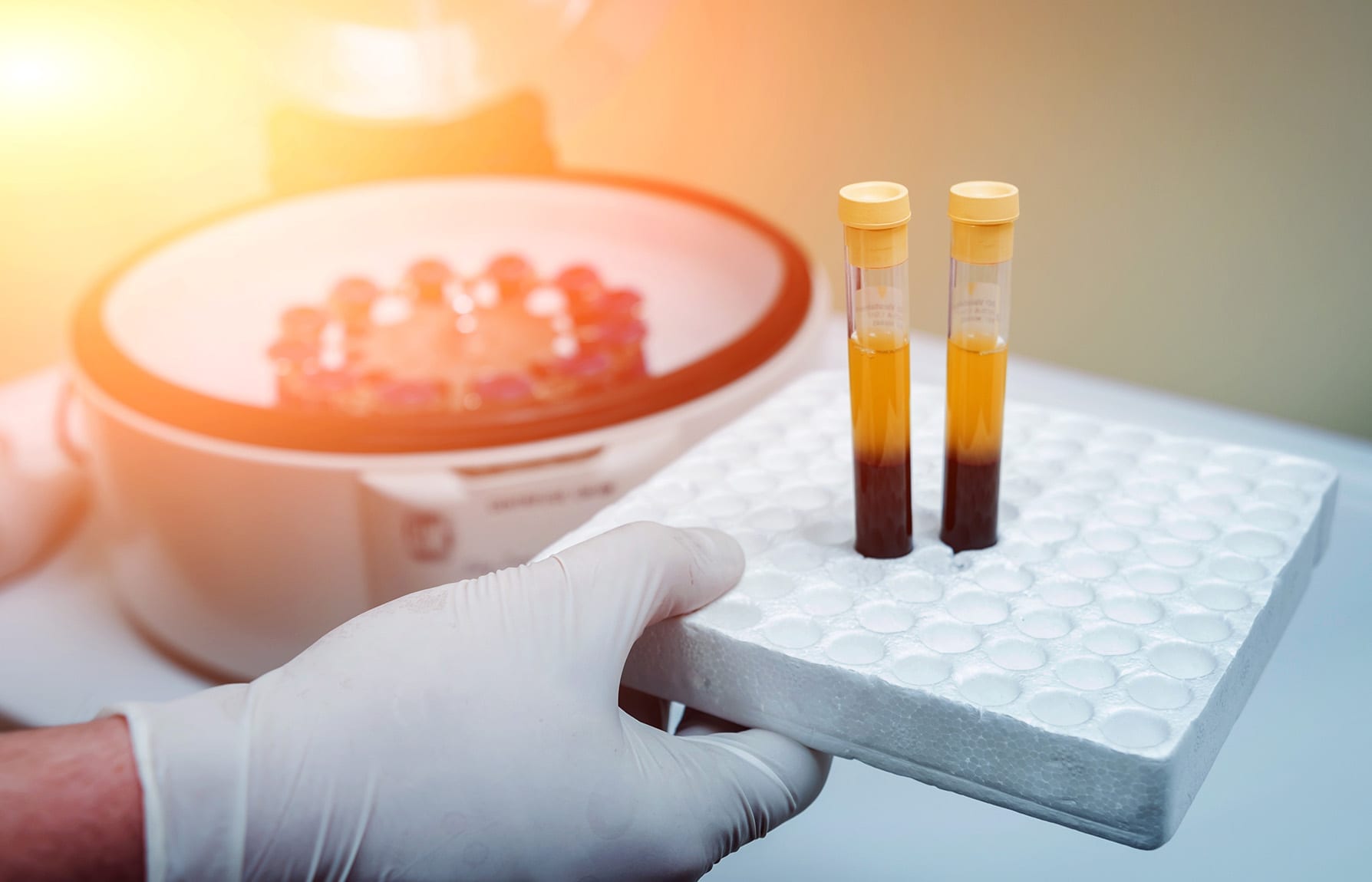
4. What to Do When Isolating Cells?
Cell isolation is a complex and specific process for which you should always consult and follow an appropriate protocol. Here, we highlight some handy tips for general good practice in handling and isolating blood cells.
Avoiding hemolysis and clot formation are crucial precautions when handling blood for cell isolation. All buffers and gradients used in the process should be temperate to prevent heat shock of the sample.
Making sure the density gradient is correct, for example, 1.077g/ml in the case of Peripheral Blood Mononuclear Cells (PBMCs) isolation, will spare losing the sample due to incorrect density separation. Again, in the context of PBMCs, buffer solutions used for dilution and washing such as PBS must be calcium and magnesium-free. The presence of these two ions could activate immune cells and launch their differentiation.
As freeze-thaw cycles lyse cells and cause nuclease release, it is crucial to avoid them as much as possible in the context of preserving frozen cells integrity and proper biological functionality.
And last, but not least, it is important to note that results and how blood samples "behave" are likely to differ between healthy subjects and patients with infectious or oncological diseases.
5. What to Do When Isolating Plasma?
Although easier than cell isolation, plasma purification must be done with caution to ensure the good quality of the molecular targets.
The most crucial point for plasma retrieval is to avoid clot formation. This is done by gently shaking up and down the tube to allow sufficient mixing with the anticoagulant agents. Keeping samples cooled at approximately 4°C is additional help against clot formation and ensures good preservation of the key molecules. The optimal transport and storage temperature for blood samples intended for plasma retrieval is also set to 4°C.
Storage and freeze-thaw risks are less of an issue since there are no biologically active plasma sample components. However, best practices still advise avoiding such events. On the other hand, an important issue affecting plasma samples is hemolysis. This event corresponds to the disruption of red blood cells and the release of hemoglobin and other components into the liquid phase changing its color to pink or red.
Conclusion
Ensuring uniformity and quality of biospecimens is crucial for the successful development of "bench to bedside" solutions for patients in need. Two factors play a critical role in this process. First, use established best practices guidelines and protocols for standardized collection of main classes of blood-derived biospecimens. Second, when using a specific custom protocol, strictly respect its details to protect samples integrity and ensure they meet the study's downstream analytical purposes.
At Audubon Bioscience, we have the needed expertise to support you in your blood specimen collection project. If you wish to learn more about blood-derived products or need a partner for your custom collection, visit our peripheral blood products page or get in touch with our team!
References:
- Meddeb, R., Pisareva, E. & Thierry, A. R. Guidelines for the Preanalytical Conditions for Analyzing Circulating Cell-Free DNA. Clin. Chem. 65, 623–633 (2019).
- Gottfried-Blackmore, A. et al. Effects of processing conditions on stability of immune analytes in human blood. Sci. Rep. 10, 17328 (2020).
- Betsou, F., Gaignaux, A., Ammerlaan, W., Norris, P. J. & Stone, M. Biospecimen Science of Blood for Peripheral Blood Mononuclear Cell (PBMC) Functional Applications. Curr. Pathobiol. Rep. 7, 17–27 (2019).
- Tuck, M. K. et al. Standard operating procedures for serum and plasma collection. J Proteome Res 8, 113–117 (2010).

